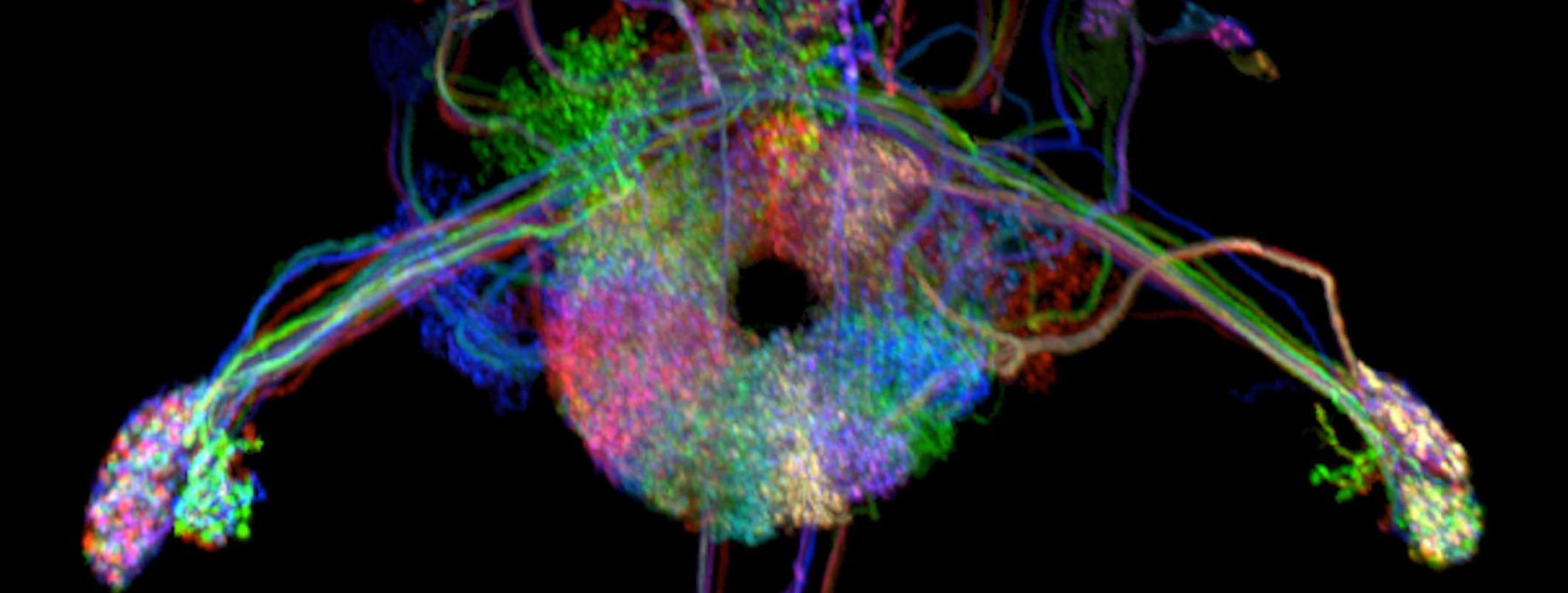
We pursue a mechanistic understanding of how populations of neurons interact to process sensory information and guide behavior. We record the activity of entire populations of one or more genetically identified types of neurons in the fly, Drosophila melanogaster, to acquire a complete view of neural dynamics in diverse behavioral contexts. Such experiments represent a new opportunity to understand how information is transformed, how it diverges across different populations of neurons, and how these processes collectively achieve behavioral goals. It reveals how simple computational principles enable coherent interactions, or sometimes resolves conflicts, among brain areas.
We develop theoretical and computational models to explain physiologically observed neural activity. We rigorously test predictions of such theories and models in behaving animals in a virtual reality arena, using a variety of physiological techniques, including genetically targeted two-photon calcium imaging and optogenetic perturbation. In this way, we aim to ultimately build a robust, physiologically tested, principle-based models of brain computations that may explain how animals flexibly interact with the surrounding world.

Techniques
Two-photon Imaging
We combine virtual reality and physiology. A tethered fly is allowed to walk on a ball or fly, while its behavior is monitored. We measure the rotation of the ball, or measure the difference between fly's left and right wingbeat amplitudes and use this motor information to let the fly control the rotational velocity of the visual environment. We call this a closed-loop experiment because we close the loop from the fly’s motor output to the visual scene that the fly experiences. We express the genetically encoded calcium indicator, GCaMP, in a genetically identified set of neurons to monitor calcium activity using a two-photon laser scanning microscope.
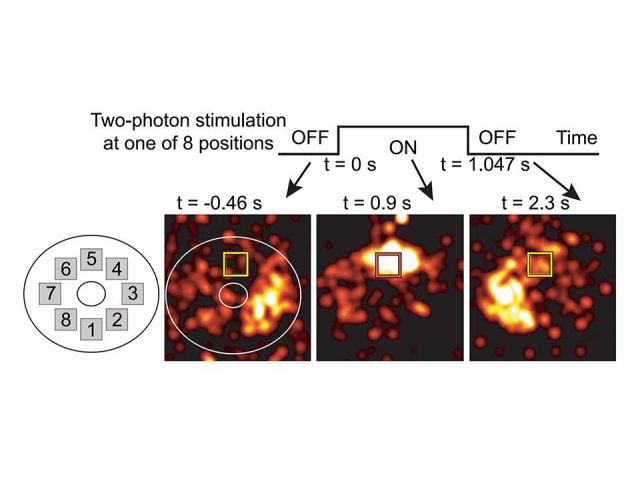
Optogenetics
We express a channelrhodopsin variant and a calcium indicator in a genetically identified set of neurons. Only a small subset of neurons is stimulated while the activity of the entire population is recorded. To do this, we developed a technique allowing simultaneous imaging and stimulation.
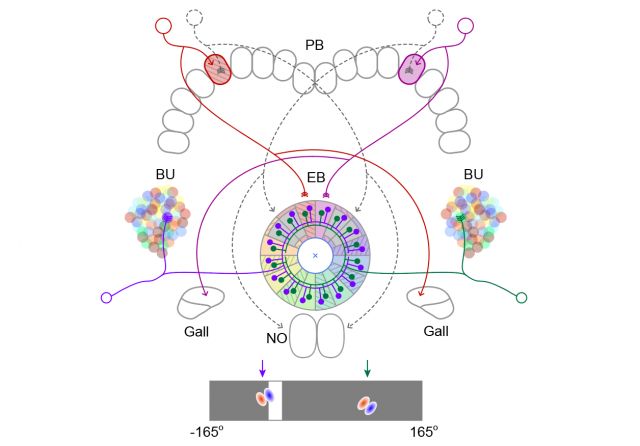
Computational Modeling
A fundamental understanding of computational principles underlying complex neural dynamics does not come just from phenomenology, but from quantitative formulation. We develop dynamical models of the fly's navigational system and test their predictions using physiological tools such as two-photon imaging and optogenetics.